The Effects of Groundwater Pumping on Natural Spring Communities in Owens Valley[1]
Deborah J. Perkins, Bruce N. Carlsen, Mike Fredstrom, Richard H. Miller, Cindy M. Rofer, Gregory T. Ruggerone, and Carolyn S. Zimmerman[2]
Abstract.—Attempts to return the marsh at Little Black Rock Spring to its pre-pumping condition have been unsuccessful. Runoff water does not adequately replace spring water in nutrient and groundwater levels. The lack of mitigation for water loss due to pumping the Springfield and Fish Springs Lake has resulted in the total loss of marsh habitats.
General Introduction
The purpose of this study was to determine the changes in water chemistry and plant and rodent distribution as a result of groundwater pumping at three historical spring sites. Since none of the affected sites were studied before pumping began, comparisons between affected and unaffected sites were made to estimate the extent of change that had occurred. The first section deals with Little Black Rock Spring as the subject of a mitigation effort to reestablish the marsh community lost as a result of nearby pumping. Water diverted from mountain runoff was introduced to the area to serve as a replacement for natural spring water. In this section, the water chemistries of a nearby well and two natural springs are compared with that of the diversion channel in an attempt to determine the differences between natural spring water and diverted water. The distributions of aquatic and marsh plants and rodents are included in the comparison. The second section compares the plant community structure in two former springs that have dried up due to water removal with that of a natural, free-flowing spring. The importance of vegetation structure to rodent distribution is also discussed.
Study Sites
Little Black Rock Spring
Little Black Rock Spring is located in Inyo County, California, approximately 14.5 km. (9 mi.) north of Independence and 1.6 km. (1 mi.) east of US 395. Prior to groundwater pumping, the spring flowed from the base of a fractured basaltic lava flow down a hill to a basin containing several ponds. Percolation from the ponds was minimal because the natural water table was high, approximately 1.0–2.4 m. (3–8 ft.) below ground level [Lee 1912]. The aquifer that supplied the spring was assumed to supply a nearby well, #351, water from which was used to estimate the chemical properties of the natural spring water at Little Black Rock Spring.
Groundwater pumping began in the early 1970s to supply water to the Little Black Rock Springs Fish Hatchery, because the volume of spring water was declining. Soon after pumping began, the spring ceased to flow. Water was then supplied to the spring area by Black Rock Ditch (fig. 1) which received water from two sources. The major source was Division Creek, a system of concretelined ditches and natural channels which receives water from Scotty Spring at the base of the Sierras. The second source was water diverted from Goodale Creek. Black Rock Ditch received water from the creek only when the damming action above the ditch intake caused an overflow; this seldom occurred.
Black Rock Ditch (site 1 for water chemistry) was a natural channel roughly 3–5 m. (10–15 ft.) wide and 0.6–0.9 m. (2–3 ft.) deep; it was
[1] Paper presented at the California Riparian Systems Conference. [University of California, Davis, September 17–19, 1981.]
[2] Deborah J. Perkins is Master's Candidate, California State University, San Diego. Bruce N. Carlsen received his B.S. from the University of California, Santa Barbara. Mike Fredstrom will receive his B.S. from California State University, Fullerton. Richard H. Miller is Master's Candidate, University of Utah, Salt Lake City. Cindy M. Rofer is Master's Candidate, Humboldt State University, Arcata, Calif. Gregory T. Ruggerone is Master's Candidate, University of Washington, Seattle. Carolyn S. Zimmerman is attending Arizona State University, Tempe.

Figure 1.
Black Rock Ditch which supplies water
to the Little Black Rock Spring area.
lined with willows and other woody and herbaceous plants. Water flowed from the channel to the main pond (site 3) through a 12-in. diameter pipe. The main pond averaged 15 cm. in depth, contained at least three species of green algae, and was surrounded by three-square rush (Scirpusamericanus ) and the cat-tail Typhadomingensis . A loose rock dam below the intake maintained a water level high enough to allow flow into the pipe. From the pipe, water flowed through a small channel and into the pond system (fig. 2). On occasion the rock dam deteriorated to the point that water could not reach the intake pipe. This situation occurred during our study and resulted in a significant drop in water level in the ponds from evaporation and percolation. During our study, the water table was measured at 11.3–11.5 m. (37–38 ft.) in a nearby observation well.

Figure 2.
Main pond and marsh area at Little Black Rock Spring
with Anemopsis californica in the immediate foreground.
Fish Springs Lake
Fish Springs is located 8 km. (5 mi.) south of Big Pine (Inyo County) and 0.4 km (0.25 mi.) west of US 395. It is the site of a fish hatchery run by the California Department of Fish and Game (DFG). Fish Springs Lake, the study site, is located several hundred meters south of the hatchery. The lake once occupied a depression below the face of a vesicular basaltic lava flow. Caliche deposits at the site suggest that the water level may have been 0.6–0.9 m. (2–3 ft.) deep at one time (fig. 3). Pumping at the fish hatchery was increased in 1971 to replace the dropping volume of spring flow. Flow from Fish Springs ceased in 1971 (Williams 1978), which corresponded with the increase in pumping at the hatchery. It is likely that the lake and the spring received water from the aquifer associated with the fractured lava flow. This source is not established due to lack of data at the study site. The lake has been noted as having water some years and being dry in others. The amount of water in recent years may be attributed to the accumulation of runoff.

Figure 3.
Fish Springs Lake, a dry bed covered with
dead reeds. Note caliche deposits on rocks.
The Springfield
The Springfield is located 2.4 km. (1.5 mi.) east of Independence. The topography is essentially flat with the exception of broad, shallow

Figure 4.
Rabbit-brush (Chrysothamnus nauseosus ),
the dominant plant at the Springfield.
depressions. The spring area had been invaded by the desert shrub rabbit-brush (fig. 4). The five wells adjacent to the site pumped water from several different levels of a complex layering of sand, clay, and gravel. The deepest well pumped from 143 m. (468 ft.). Another well had artesian flow until 1971, after which the depth was approximately 4.6–7.6 m. (15–25 ft.). Pumping began in the early 1900s, which made this area the study site with the longest pumping history. A brush fire passed through the area in 1971 and destroyed most trees and marsh vegetation.
Fish Slough
Fish Slough is located 8 km. (5 mi.) north of Bishop in the southernmost extension of the volcanic tablelands. The slough is a marsh and pond area that extends 6.4 km. (4 mi.) back toward Bishop and empties into the Owens River. There are three primary springs within the slough, the northernmost of which is site 1. This spring issues near the base of a steep slope of volcanic tuff inside a circular pool constructed of rocks and cement. Overflow continues south through a system of channels and ponds into which the other springs flow. Site 3 was a pond located 4 km. (2.4 mi.) south of site 1. The pond was surrounded by three-square rush, and the bottom covered with a thick mat of macrophytes. The slough is bounded on the east by a faulted block of Bishop Tuff. Spring flow at the slough might have been caused by the impoundment of water from the drainage basin at the fault adjacent to it. If this was the case, then agricultural drainage and precipitation in the basin were major sources of spring flow at the slough.
Collins Warm Springs
Collins Warm Springs is located 13 km. (8 mi.) north of Big Pine and 8.6 km. (5.5 mi.) east of US 395 at the base of the Inyo/White Mountains. It has been designated part of the Owens Valley Pupfish Sanctuary. Springs issue from several sources at the base of a weathered and fractured marble outcrop that was probably exposed by faulting. The main group of springs (site 1) flows into a man-made pond (site 2), which was dammed to support the pupfish population. The pond was 0.9 m. (3 ft.) deep, and the bottom was partially covered with Oscillatoria sp. Water flowed from the pond through a pipe down to an extensive marsh area. Except for the building of the dam, the area had been undisturbed for some time.
Analysis of Mitigation Success
Introduction
The purpose of this section is to analyze the success of mitigation at Little Black Rock Spring. The spring area has undergone significant biological changes since the natural spring flow ceased. Before cessation of spring flow, flatworms (Dugesia or Planaria ) were observed living near the spring source; other native aquatic organisms that require flowing spring water were also present.[3] The wetland plant yerba mansa (Anemopsiscalifornica ), which is known to thrive in unstable water conditions, was not a dominant plant in the basin.[4] When well #351 was drilled in the early 1970s, the water table dropped to the point that spring flow ceased. During the period when no water entered the basin, both aquatic and riparian plants were affected. The basin which lay directly beneath the spring lost large sections of vegetation, so that only bush stumps and dry grass clumps remained. In comparison to a 1947 aerial photograph of the area, the marsh was roughly one-third of its pre-pumping size. Yerba mansa became a dominant plant in the remaining marsh area.
No data were available on the pre-pumping condition of Little Black Rock Spring. In lieu of a direct comparison between pre- and postpumping conditions, an indirect comparison was made. The water chemistry of well #351 was considered the closest to that of the original spring water. Well #351 and two natural springs, Fish Slough and Warm Springs, were used for the water chemistry comparison. Plant and rodent distributions at Little Black Rock Spring and Fish Slough were compared in an attempt to estimate the magnitude of changes that had occurred at Little Black Rock Spring since its source of water had changed.
Four aquatic plants were considered indicator species for dissolved ion levels. By comparing the ion levels at well #351 and Fish Slough, the potential for certain aquatic plants at the old Little Black Rock Spring could be established. The key word is "potential" since the composition of the pre-pumping plant community was unknown. Three assumptions were made regarding the plant species found at Little Black Rock Springs. 1) The plant species present following the transition were those able to tolerate the changes in water chemistry and availability. 2) New species had probably not had the time or the means to invade the area. 3) The absence of certain species could be explained either by their recent disappearance or by their never having been introduced.
Potamogetoncrispus , a rooted vascular plant, showed optimal growth at 50 m g nitrogen per l. and 50 m g phosphorus per l. when grown in greenhouses (Mulligan and Baranowski 1969). More recently Mulligan etal . (1976) found that Potamogeton sp. grew well under conditions of high fertility (75,000 m g nitrogen per l. and 75,000 m g phosphorus per l.) in pond experiments. P . pectinatus cannot tolerate waters with a conductivity below 200 m mhos (Seddon
[3] Larson, E. 1977. Personal conversation. High school biology teacher, Bishop, Calif.
[4] DeDecker, M. 1977. Personal conversation. Local field botanist, Independence, Calif.
1972). Another rooted plant, aquatic buttercup (Ranunculusaquatilis ), has a tolerance to low conductivity and can be found in waters as low as 60 m mhos.
Lemna is a genus of floating plants which must absorb all of its nutrients from the water. In the laboratory, Hicks (1932) studied the growth of Lemnaceae in buffered media lacking trace metals and set the pH range at from 4.5 to 8.0. McLay (1976) broadened the pH range for Lemna spp. to from 4 to 10, with optimal growth at pH 6.2 He used these results to support his findings in the field that the growth of Lemna perpusilla was poor at pH 8.23 (McLay 1974).
Littlefield and Forsberg (1965) found that Chara sp. absorbed phosphorus equally well through all of its parts, which indicates that water nutrients could have a profound effect on the distribution of Chara sp. It has been found that Chara sp. exists in waters that contain 20 m g total phosphorus per l. or less, of which dissolved orthophosphates constitute a very small portion. A tolerance limit of Chara sp. to orthophosphates was reported by Forsberg (1965) to be 1 m g PO4 phosphorus per l. or less. The exclusion of Chara sp. was observed by Mulligan etal . (1976) by fertilizing experimental observation ponds. Chara succession was described by Wood (1950) and Crawford (1977), where three species of Chara and some Najasflexilis succeeded to a community of Najas , Potamogeton , and Typha in three years. The average alkalinity of waters in which Chara spp. are found was 2.55 milliequivalents per liter (Forsberg 1965), but some species were ubiquitous (Spence 1967).
According to Smith (1974), the alteration of one component in an ecosystem, such as lowered groundwater levels, often leads to changes in the community as a whole. Several ecological indices were used to estimate the degree of change in the distribution of marsh plants at Little Black Rock Spring. Species diversity, which combined the density and relative importance of all species found in a given area, was compared between sites. The assumption in this comparison was that undisturbed marshes in the area were expected to have comparable diversities, such that changes in the plant community at Little Black Rock Spring would be reflected in the species diversity of the site. Smith states that a greater number of vegetative life forms indicates a more complex ecosystem and that the biomass of total plant cover largely determines food resource availability. The number of life forms, as well as total plant cover, were compared between sites in an attempt to further delineate differences in the plant community and potential animal community between sites.
Within a freshwater marsh ecosystem, very slight alterations in environmental parameters often result in major changes in the riparian flora (Mason 1957). Typha domingensis , the common cattail, is classified as an early successional species (ibid .) and typically will invade newly flooded areas. Low salinity levels provide an advantage to the germination of Typha seeds over those of rushes (Scirpus spp.). Water salinity is a major determinant in the distribution of marsh vegetation (ibid .). The accumulation of organic matter is also important to distributions in that it is a necessary factor in the succession to other species (Spence 1967), such as those in the rush family.
Changes in food and water availability (Meserve 1974) and vegetation structure (Rosenzweig 1973; Beatley 1976) have been found to affect rodent populations. When a perturbation of water availability causes local habitats to change, the abundance of rodents, which are habitat specialists, is likely to change. Many rodent species seem to prefer one microhabitat over another (Price 1978). It has been suggested that body size and morphology determine which microhabitat is appropriate to maximize the seed collecting efficiency of a heteromyid (ibid ). Each species is most dense where its preferred microhabitat is abundant (ibid .). Experimental augmentation of one microhabitat led to the increase in density of the appropriate microhabitat specialist. Thus, the availability of an appropriate microhabitat determines species abundance on a local scale (ibid .).
However, physical adaptation is not the only means of allocating separate microhabitats. Interspecific competition within a foraging habitat (ibid .) allows different species to coexist in the same area. Price has shown that removal or addition of a competitor results in a predictable shift in resource use by other species. For example, when Dipodomysmerriami was removed from its open habitat, Perognathusamplus shifted its distribution to the open habitat (Wondolleck 1978). D . microps (a leaf eater) was replaced by D . merriami (a seed eater) when a disturbance reduced the amount of vegetation cover (Beatley 1976). Schroder and Rosenzweig (1975) found that two species of Dipodomys avoided competition by habitat selection. Cricetid rodents also compete for space (Crowell and Pimm 1976). Cricetid species with similar nutritional requirements also appear to coexist through the use of habitat selection (M'Closkey and Fieldwick 1975).
The reasons for selecting one microhabitat over another have been a subject of controversy for years. Originally, rodents were believed to partition food resources by seed size selection. More recently, rodents have been shown to collect different sizes of seeds whether they forage in the same habitat or not (M'Closkey 1980). M'Closkey went on to state that the seed sizes collected by one species of rodent are determined by the preferred microhabitat of that species, such that two of the elements involved in allowing heteromyid coexistence are reduced to one dimension—microhabitat selection. In addition, Rosenzweig and Winakur (1969) found that plant density, soil type, and bush height are all important para-
meters in habitat selection by Dipodomys spp. Coexistence is probably maintained by a balance between habitat selection with its many parameters and interspecific competition.
Methods
Water Chemistry
Water samples were collected from Fish Slough, Little Black Rock Spring, well #351, and Warm Springs using a horizontal Van Dorn bottle. All filters and sample bottles were acid washed and rinsed with 10 megaohm water. In the field, concentrated sulfuric acid was added to the nitrate samples until pH 2 was reached. Then the nitrate, nitrite, and phosphorus samples were put into a deep freeze until analyzed. Nitrite, reactive phosphorus, and dissolved oxygen were analyzed using methods from Strickland and Parsons (1972). Nitrate was determined by the brucine method (American Public Health Association 1965). Field pH and alkalinity measurements were made with a Perkin-Elmer model Coleman 37A pH meter. Lab measurements were made with a Corning model 10 pH meter. Alkalinity titrations were done with concentrated HC1 to a pH of 4.5. Specific conductance, which is directly proportional to the concentration of dissolved salts, was measured with a Lab-Line Lectro Mho-meter model MC-1 Mark IV.
Vegetation Sampling
The marshes were quite patchy in distribution and the methods employed in this study produced data that could be used only to approximate plant distributions. The line transect method (Smith 1966) was used to describe the plant community inhabiting the dried marsh at Little Black Rock Spring. Sixteen lines, 25 m. long, were placed at 10-m. intervals parallel to the old spring source. A random quadrat system was used in the area adjacent to standing water at Little Black Rock Spring and Fish Slough. Mean cover and species importance were calculated using techniques described by Mueller-Dombois and Ellenberg (1974). Species diversity was calculated using the Shannon-Wiener index (Krebs 1972). Plant species were divided into the following five life form categories: aquatic sedge; alkali grass and reed; alkali herb; shrub; and tree. The plant community within standing water was described from observations. Vegetation and terrain were characterized at Fish Slough by recording the three to five most dominant plant species and percent plant cover.
The effects of water table depth and some soil variables were estimated from the data gathered at 21 random grid points at each site. The grids were of equal area (100 m. × 70 m.) and were placed adjacent to the water channel at each site. The grid points were placed at 10-m. intervals and served as the loci of 1 sq. m. quadrats where soil was sampled. A water extraction pressure plate was used to determine soil moistureholding capacity. Soil filtrates were measured for conductivity with a Yellow Springs model 31 Conductivity Bridge and pH with Hyrion pH paper.
Rodent Sampling
Rodents were captured in Sherman live-traps. Each study site was trapped for four consecutive nights and later for two consecutive nights. Trapping was adjusted to the lunar cycle so that each site had the same approximate amount of moonlight during its trapping period (Lochard and Owings 1974). The traps were placed on line transects with the traps 10 m. apart, 20–30 traps per transect, and three to five transects per site. The transects were arranged radially from the spring site.
Results
The water chemistry data presented in this paper were limited to those sites in each area that were located at the spring source (site 1) and the first major pond (sites 2 or 3). The natural spring waters at Fish Slough and Warm Springs had a higher alkalinity and specific conductivity than Little Black Rock Spring (table 1). Orthophosphates and nitrates were much higher at Fish Slough and well #351 than at Little Black Rock Spring (table 2). The data from Warm Springs showed that each spring had unique characteristics. However, the significant comparison lay between well #351 and Little Black Rock Spring. The water which originally supplied the spring was very different from the water that came from Black Rock Ditch. Orthophosphates had increased at site 3 in Fish Slough from <.004 m g PO4 , phosphorus per l. in 1973 to 4.70 to 10.23 m g PO4 phosphorus per l. during the summer of 1978.[4]
| |||||||||||||||||||||||||||||||||||||||||||||
Figure 5 shows that diurnal water temperatures were more constant at the water source, which corresponded to site 1 at Fish Slough and Little Black Rock Spring. In both cases, site 3 was a pond where the flow rate had decreased; the
| ||||||||||||||||||||||||||||||||||||
water temperature increased during the day. The temperature change at Little Black Rock Spring was more drastic and required a greater range of tolerance from all aquatic organisms.
At Little Black Rock Spring and Fish Slough, site 1 showed little fluctuation in oxygen saturation (fig. 6 and 7). This indicated that the oxygen input from the water source was constant and there was little photosynthetic activity. In both cases, site 3 had a larger fluctuation due to the large standing crop of primary producers associated with each pond. Site 3 also had the largest change in pH, presumably due to the higher photosynthetic rate. The primary productivity of Fish Slough was higher at its peak than Little Black Rock Spring (185% versus 132% saturation). The slight increase in saturated oxygen in Black Rock Ditch (site 1) came from the macrophytes that lined the ditch.
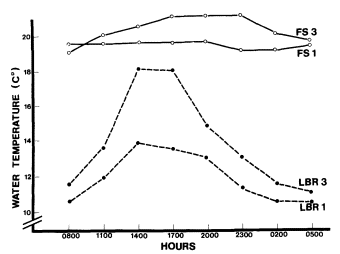
Figure 5.
Diurnal water temperature (°C) for sites 1 and
3 at Fish Slough and Little Black Rock Spring.
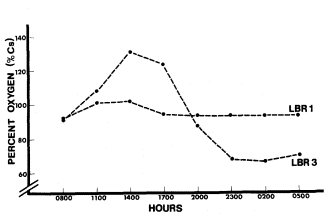
Figure 6.
Little Black Rock Spring diel oxygen study.
(Note that the drop in oxygen before sunset
resulted from a sudden cloud cover.)
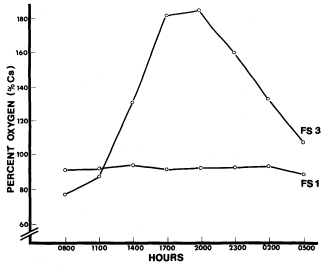
Figure 7.
Fish Slough diel oxygen study.
According to all of the community indices that were calculated, the dried marsh of Little Black Rock Spring has undergone marked changes (table 3). The number of species present, species diversity, total cover, and number of life form categories were much lower. The marsh that survived the transition was slightly different than the Fish Slough marsh in that fewer species were present at Little Black Rock Spring, causing species diversity to decrease slightly.
| |||||||||||||||||||||||||
In comparing marsh areas, the dominant species were found to be quite different (table 4). The dominant plant at Fish Slough was a sedge, Eleocharisparishii . The most abundant herb was Glycyrrhizalepidota , which covered less than half of the area of E . parishii . At Little Black Rock Spring, the most dominant plant was the invading herb Anemopsiscalifornica , while E . parishii covered one-third of the area of A . californica . Another species of interest was wire grass (Juncus balticus ), which had the same approximate cover area at both sites. Both Scirpusamericana and Typha domingensis were present in the marsh at Little Black Rock Spring, but not at Fish Slough.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Unquantified observations of the entire marsh and pond areas revealed stands of marsh plants that were not sampled by the grids. Most of the waterways at Fish Slough were lined with dense stands of Scirpusamericana , while Typha domingensis was sparse. In contrast, pure stands of T . domingensis were mixed with stands of S . americana at Little Black Rock Spring. The pond (site 3) at Fish Slough contained several macrophytes that had formed a mat on the floor of the pond. Of those species, the ones that were considered to be indicative of certain water characteristics were Najas marina, Potamogetonpectinatus , and Chara sp. At Little Black Rock Spring, the little channel that carried water from the source to the ponds contained Ranunculusaquatilis and Lemna sp. The main pond at Little Black Rock Spring (site 3) did not contain the same species as did site 3 at Fish Slough.
Some of the soil parameters were averaged for each site. Mean soil conductivity at Little Black Rock Spring was 616 mmhos, while Fish Slough had 2,717 mmhos. Mean soil pH was 6.6 at Little Black Rock Spring and 5.7 at Fish Slough. Soil moisture-holding capacity decreased linearly with increasing distance from the channel at Little Black Rock Spring (fig. 8). At Fish Slough, soil moisture-holding capacity was not dependent on distance from the channel. Depth to the water table at Fish Slough varied from 0.5 to 1.0 m. at a distance of 0–50 m. from the water channel. At Little Black Rock Spring, where the natural water table was at approximately 11.5 m., the artificial water table from the pond dropped off quickly. At the edge of the pond, the depth to water had dropped to the lowest limit of the soil auger, 1.8 m. At 20–50 m. from the channel, the water table was below 1.8 m.
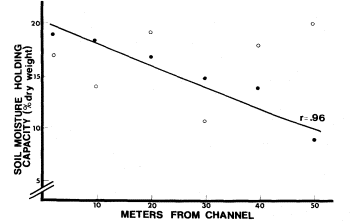
Figure 8.
Soil moisture-holding capacity as it varies with distance from the
water channel. o—Fish Slough, o—Little Black Rock Spring.
The plant species present on the soil sample grids reflected the water table depth. On the line closest to the channel at Little Black Rock Spring, wire grass was abundant. Twenty meters away from the channel, drought-tolerant plants like rabbit-brush, Sporobolusairoides , and salt grass (Distichlisspicata ) were present. At 50 m., Little Black Rock Spring had sparse vegetation cover consisting of rabbit-brush and
annual weeds. At Fish Slough the plants found 20 m. from the channel were Eleocharisparishii , Juncus balticus , and Glycyrrhizalepidota . At 50 m. the marsh plants were still abundant.
The rodent species common to both areas were the Heteromyids Dipodomysmicrops , D . ordii , and Perognathusformosus , and the Cricetids Neotomalepida , Peromyscusmaniculatus , P . boylii , P . crinitus , and Reithrodontomysmegalotis . The additional species found at Fish Slough was D . merriami , for a total of nine species. Little Black Rock Spring had three additional species, Neotomafuscipes , Onychomystorridus , and Peromyscustruei , for a total of eleven species.
Five hundred sixty-eight trap nights were used to calculate the rodent-habitat associations. These were considered preliminary results, since many more trap nights would be needed to make conclusive correlations. Positive associations were made with each rodent species and, in several cases, habitat overlaps existed between species (table 5). One of the patterns that emerged from these data was that none of the Peromyscus spp. overlapped. R . megalotis was found only in closed habitats where grass and tall plants were intermixed. Dipodomys merriami and D . ordii were found in different habitats, while D . microps shared the habitats of the other Dipodomys species.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
A comparison of the chemical aspects of Fish Slough, Little Black Rock Spring, well #351, and Warm Springs showed that the water supporting the post-pumping marsh at Little Black Rock Spring was quite different from the water in the local aquifer and other natural springs. Alkalinity, salinity, and nutrients needed for primary production were all lower in the water supplied to the Little Black Rock Spring marsh from Black Rock Ditch. The low conductivity enabled Ranunculusaquatilis to survive there. The low orthophosphate level would be suitable for Chara sp., though it was not found at Little Black Rock Spring. Since the orthophosphate content of well #351 was extremely high, it is likely that Chara was not present before pumping began. Chara was not abundant at Fish Slough and might have been on the decline due to the recent increase in orthophosphates. The work done by Mulligan etal . (1976) showed that Chara could be excluded with the addition of fertilizer. The successional pattern where a Chara -Najas community changed to a Najas -Potamogeton -Typha community observed by Woods (1950) and Crawford (1977) appeared to be in progress also. The increased agriculture in the drainage basin of Fish Slough could be affecting the ecological system of the slough.
The absence of Lemna sp. in the ponds (both site 3s) was predictable because of the high daytime pH. Lemna sp. was found in the intake channel at Little Black Rock Spring, which was well protected by tall brush, and probably survived there because shading prevented a high photosynthetic rate and the accompanying increase in pH. The absence of Potamogetonpectinatus at Little Black Rock Spring was also predictable because of the low conductivity there. P . pectinatus was found at Fish Slough where the conductivity was above the lower limit found by Seddon (1972). The conductivity of well #351 was probably above its lower limit too, since the alkalinity of well #351 and Fish Slough was similar. Considering these facts, P . pectinatus may have been present before pumping and eliminated when the water source changed.
Primary production was higher at Fish Slough as demonstrated by the percent saturation of oxygen. The combination of more constant diurnal temperatures, a more constant water source, and higher nutrient concentrations probably supported more photosynthesis at Fish Slough than at Little Black Rock Spring.
The distribution of two major marsh plants, Scirpus americana and Typhadomingensis , was considerably different between the two areas and was probably due to the decrease in salinity at Little Black Rock Spring. Since T . domingensis had been classified as an early successional species which germinates easily and thrives in water of low salinity, its high abundance at Little Black Rock Spring was predictable. S . americana dominated the marsh at Fish Slough, a well-established marsh with a higher organic content in the soil and more saline water.
The marsh area at Little Black Rock Spring appeared to have sustained significant losses in area and, possibly, species richness. Before pumping, the water table was close to the surface and supported an extensive marsh. After pumping, the marsh ended abruptly at the edge of the pond where the water table dropped. Alkali deposits, remains of dead vegetation, and aerial photo-
graphy supported the theory that the marsh once extended into the area later occupied by xeric vegetation. Species richness, diversity, and life form categories were lower in the desert area, which indicates that the losses occurred during the transition and that the surviving species were forced to retreat to the remaining marsh. Actual losses and gains in species composition at Little Black Rock Spring could not be estimated from our data. However, Little Black Rock Spring supported seven fewer species than Fish Slough within the sample grids. It was concluded that either species richness had always been lower there or species were actually lost during the transition.
The rodents at Fish Slough appeared to prefer different microhabitats and demonstrated possible rodent-habitat associations. For those species that appeared to be using the same microhabitat, other resource partitioning mechanisms might have been operating, although these mechanisms were not pursued further. Habitat preferences were determined only at Fish Slough, but were considered applicable to both sites.
More specifically, Dipodomysmerriami and D . microps demonstrated the type of coexistence described in the literature. Within their common habitat, coexistence was possible because of their different food preferences. Another Heteromyid, Perognathusformosus , preferred a different habitat from the other Heteromyids. Each of the three Peromyscus species appeared to prefer a different habitat. Reithrodontomysmegalotis was found only in wet places. Each species preferred one or two habitats. It has not been determined whether these habitats were preferred as a result of competitive exclusion or a physiological need to associate with that habitat.
It is clear, however, that changes in the number and type of habitats would influence the rodents that depend on them. In the case of Little Black Rock Spring, the mitigation to replace lost water maintained a set of moist habitats which probably supported most of the original species. The differences in species between the two areas could not be linked directly to the transition to ditch water since a similar census was not performed before pumping began. The differences in species present and species richness was attributed to artificial juxtaposition of desert and marsh, such that a transect that spanned the two major habitats produced a higher species composition than a transect that crossed the more homogeneous area of Fish Slough.
Conclusion
The water diverted into Little Black Rock Spring contained significantly lower concentrations of the components that influenced the structure of the biological community. The original spring probably had higher concentrations, as indicated by a nearby well. Comparison of the post-pumping marsh to an unaffected marsh showed definite differences in the water chemistries and biological community. The low salinity at Little Black Rock Spring probably was the cause of the early successional marsh and the absence of certain aquatic plants. Low orthophosphate and nitrogen levels were probably the cause of the comparatively low primary productivity rates. The drop in groundwater and sharp percolation rate from the ponds caused an extensive marsh to evolve into a markedly dichotomous plant community. The most dominant plant species in the marsh were not the same as those in Fish Slough and were probably typical of the new habitat created after pumping began. The rodent community reflected some of the overall change, but the presence of water and moist habitats appeared to maintain a comparatively diverse population.
Estimation of Biological Changes
Introduction
The purpose of this section is to estimate some of the biological changes that resulted from unmitigated pumping in the vicinity of two natural spring areas. Fish Slough was used again as a source of comparison for changes in vegetation and rodent distributions. Warm Springs was also used for one comparison. The Springfield, once an artesian spring field and marsh, had been pumped for many years. Drought-tolerant shrubs and annuals had invaded the entire area. The observation that patches of these shrubs were dying indicated the need for a water stress study to determine whether these plants were mesic and would be affected by further reductions in the water table. Fish Springs Lake was considered an intermediate point between Fish Slough and the Springfield. The lake had dried out only a few years before the study, and the depression still caught surface runoff from time to time. More plant species were present there than at the Springfield, although they were mostly xeric species.
As in the previous section, the vegetation was sampled in order to estimate species richness and diversity, life form richness, and total cover. These indices were compared between the three areas to determine the vegetative differences produced by the pumping.
Plant water potential, according to Slatyer (1967) and Kramer (1969), is one of the best expressions of water deficit. Plants develop a water deficit as the soil water potential decreases (Slatyer 1967). Soil water potential was estimated from the pre-dawn measurement of plant water potential (Slatyer 1967; Merino etal . 1976). A plant was considered to be in equilibrium with the soil before it began to transpire during the day. The amount of transpiration was estimated by subtracting the pre-dawn water potential from the highest reading of the day. Pearcy (1974) found that excess water loss due to transpiration is controlled in some desert plants by stomatal closure. A decrease in water potential during the midday heat could indicate that stomatal closure has decreased the transpiration rate.
Atriplextorreyi was used to compare the water stress of plants at the Springfield and Warm Springs. In Owens Valley, it is found on the valley floor but not on the alluvial fans. The combination of this distribution and the close relation of this species to A . lentiformis , a mesophyte (ibid .), led to the hypothesis that A . torreyi is also a mesophyte. Since Merino etal . (1976) found that the annual range of plant water potential was directly associated with the annual water table depth, a mesophyte such as A . torreyi could be monitored to indicate changes in groundwater depth.
The absence of riparian systems and those defined by a more diverse vegetation structure was considered in relation to the rodent population. The first section presented the discussion on habitat preferences in rodents. Rodent homerange size and density were also considered in this study since the literature describes the dependence of those factors on vegetation density. Stickel (1960) found that Peromyscusleucopus had larger home ranges when the population density was small. She suggested that this relationship existed as a result of sparse vegetation. In a temporal study of desert rodent populations, Whitford (1976) found that most rodent species responded to fluctuations in rainfall and primary production by reductions in density during drought periods. Both Peromyscus spp. and Musmusculus responded to an increase in plant production with a relatively large population increase.
The comparison of affected and unaffected spring areas was the only method available to estimate the changes that had occurred as a result of pumping. The comparison should not be considered a statement of actual change involving a given species, but a statement of the potential change in magnitude of the physical and biological resources.
Methods
The vegetation at the Springfield and Fish Springs Lake was sampled with the line transect method (Smith 1966). At the Springfield, 10 lines 25 m. long were spaced at 10-m. intervals parallel to the spring source. Sixteen lines of similar length and orientation were placed at Fish Springs Lake. At Fish Slough, 24 random 4-sq. m. quadrats were sampled from a grid. From the data collected, mean cover, species importance (Mueller-Dombois and Ellenberg 1974), species diversity (Krebs 1972), species richness, and life form number were calculated.
The individual plants of Atriplextorreyi were sampled at the Springfield and Warm Springs by choosing those nearest to a center point on radial transects. At each reading, three twigs were cleanly severed from each of five to 10 plants. A Scholander Pressure Bomb was used to determine the xylem sap tension during three parts of the day: pre-dawn, mid-morning, and midday. Each plant was sampled on two different days at each site. The results were averaged from each time period at each site.
Rodent species lists for the Springfield, Fish Springs Lake, and Fish Slough, and habitat associations at Fish Slough were determined by the same methods described in the first section. Home range data were collected at the three sites from a 10 m. × 10 m. grid on which each intersection was a trapping station. Two Sherman live traps were set at alternate stations every evening, using mixed birdseed as bait. Traps were inspected early the next morning, and captured rodents were identified and tagged by toe clipping. Rodents were released at the station where they were captured. Traps were set for 12 nights at Fish Springs Lake and 11 nights at Fish Slough and the Springfield. The first visit consisted of four consecutive nights, and thereafter each visit was two consecutive nights. Visits to each site were alternated so that each site was trapped during each phase of the moon. During the last two trapping sessions at each site, additional traps were added to the periphery of the grid. Home range areas were calculated using the exclusive boundary method (Stickel 1954). Rodents trapped only on the periphery were not used unless their home range area was equal to or larger than those inside the grid.
Since the number of trap nights was not sufficient to establish a constant home range size, a home range rate was developed. The relative home range rate was estimated by regressing the cumulative home range area against the number of recaptures, which should give the rate at which the home range size increases with each successive capture. The logic behind this technique is that a species with a larger home range will cover more area each successive night than a species with a smaller home range. Thus, the rate of increase or the home range rate should reflect the actual home range size. The null hypothesis H ; = 0 was tested for each slope at the 5% significance level. Slopes were then compared by using the Student t-test at the 5% significance level (Zar 1974).
Results
The structure of the plant communities at the Springfield and Fish Springs Lake was quite different from that of Fish Slough (table 6). Species richness, total cover, and life form number were all lower at the pumped sites. Diversity at Fish Springs Lake was still relatively high, because all of the species were accounted for even though the majority of the individuals were dead. Diversity at the Springfield was very low because the transect lines only touched one perennial. A couple of other perennials were present nearby and the annual Chenopodiumberlandieri var. sinuatum was heavily intermixed with Chrysothamnusnauseosus , the dominant perennial. There was an obvious shift from marsh vegetation at Fish Slough (table 4) to droughttolerant species at Fish Springs Lake and the Springfield (table 7).
| |||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||
The xylem sap tensions mesured on August 10 and 11 reflected the high ambient temperature, ranging from 13–45°C at the Springfield and 16–41°C at Warm Springs, respectively. The predawn measurement, which reflected the soil water potential, was quite different between the sites (fig. 9). The water potential increased as the temperature rose, but dropped off in the afternoon. On August 24 and 25, the temperatures ranged from 10–31ºC at Warm Springs and 13–35°C at the Springfield, respectively. The soil water potential was lower for both sites, although the Springfield maintained a higher potential. The diurnal changes in water potential showed the same pattern as those on August 10 and 11.
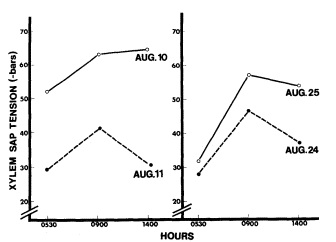
Figure 9.
Xylem sap tension at Warm Springs · and the Springfield o.
Fish Slough had nine species of rodents, while Fish Springs Lake had seven species, and the Springfield had six species. The three sites had only four species in common: Dipodomys merriami ; D . microps ; Neotoma lepida ; and Peromyscusmaniculatus . The other species found at Fish Slough are listed in the first section. Fish Springs Lake had Perognathusformosus , Peromyscusboylii , and P . crinitus . The Springfield had D . ordii and Onychomys torridus .
Habitat preferences were calculated only at Fish Slough. Those results are presented in the first section. Habitat comparisons were possible between sites because the pumped sites had such distinct differences in vegetation structure. Fish Springs Lake had only one life form category, that is, alkali grass and reed, while the Springfield had only the shrub category. Fish Slough, on the other hand, had five categories, including aquatic sedge, alkali grass and reed, alkali herb, shrub, and tree.
Peromyscusmaniculatus was the only species out of the four species in common with all three sites that had a home range rate that was significantly higher at the pumped sites (fig. 10). The lack of significance for the other species may have resulted from low capture rates.
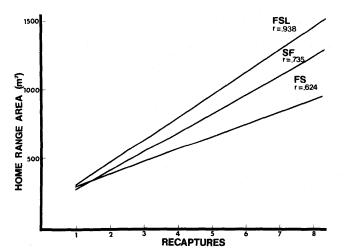
Figure 10.
Home range rates of Peromyscus manicultus at Fish Springs
Lake (FSL), the Springfield (SF), and Fish Slough (FS).
(Rejected H0 : b = 0, b1 = b2 , b2 = b 3 at p<.05).
Discussion and Conclusion
The obvious effects of unmitigated groundwater pumping on marsh vegetation are a drop in the groundwater table and a loss of surface water. Marsh vegetation was lost entirely from
Fish Spring Lake and the Springfield, and plant diversity was ultimately reduced to include only those plants that could survive on ephemeral water or deep groundwater. A mesophyte such as Atriplextorreyi was subjected to further drops in the groundwater table, such that water could become inaccessible even to long tap roots. Areas which have been pumped to that extent and beyond can be expected to lose perennial vegetation and only support those annuals that grow as a result of precipitation accumulation. Rodents that lived in pumped areas such as the Springfield could not depend on free water except that which was contained within the leaves of annuals and perennials.
Water-dependent species did not remain in an area that was pumped to the extent of losing its marsh vegetation and standing water. Rodent species that depend on seeds are most adapted to the conditions prevailing at pumped sites. Those species that are physiologically adapted to such an environment would be further limited by the low diversity of vegetation structure which is necessary for the coexistence of some species. Those individuals that do inhabit the pumped sites must finally maintain a larger home range in order to gather enough food to exist. But home range size cannot be extended indefinitely because the energy budget of each species requires a balance between the energy expended and the energy gained in foraging.
General Conclusion
The attempts to mitigate the effects of pumping at Little Black Rock Spring have resulted in a marsh community that is probably quite different from the original marsh. The combination of nutrient-poor water and a lower water table have resulted in a smaller marsh area dominated by early successional marsh species. The area once inhabited by marsh vegetation has been invaded by more drought-tolerant perennials. The rodent community is still rather diverse, but the reduced marsh and encroaching desert have probably changed the overall structure of the rodent community.
Despite the serious loss of environmental quality at sites where water removal occurred, mitigation proved better than no mitigation at all. This was demonstrated in the cases of the Springfield and Fish Springs Lake. Both the plant and rodent communities were much less diverse than those found at natural spring sites. The reduction in plant diversity and the increase in water stress at the dewatered Springfield indicated that even the desert mesophytes that live there could be endangered if the water table drops further.
Acknowledgments
This study was possible through the cooperative effort of eight people assembled by a National Science Foundation grant (SDI78-03663) in the Student Originated Studies program. The study was conducted during the summer of 1978 after seven of the participants had earned their bachelor degrees. Each participant was responsible for the experimental design and write-up of a particular subject. Bruce N. Carlsen handled the limnology section, while Mike Fredstrom wrote the hydrology section. Carolyn S. Zimmerman and Cindy M. Rofer were responsible for the vegetation sections. The rodent sections were handled by Gregory T. Ruggerone and Richard H. Miller. J. Michael Sanders assisted Deborah Perkins with a section on water stress which was not used in this report. Each person was indebted to the others for three months of assistance in his or her field research.
Dr. Richard E. MacMillen, our faculty advisor, gave us much needed advice and the room to experience the successes and pitfalls of such a study. Several residents of Owens Valley and the employees of the Los Angeles Department of Water and Power, the Caifornia Department of Fish and Game, and the Bureau of Land Management supplied important information about pre-pumping situations without which we could not have had a clear picture of the pumping issue.
Literature Cited
American Public Health Association. 1965. Standard methods for the examination of water and wastewater (twelfth edition). 769 p. American Public Health Association, New York, N.Y.
Beatley, J.C. 1976. Environments of kangaroo rats (Dipodomys ) and effects of environmental change on populations in southern Nevada. J. Mamm. 57:67–93.
Crawford, S.A. 1977. Chemical, physical, and biological changes associated with Chara succession in farm ponds. Hydrobiologica 55:209–217.
Crowell, K.L., and S.L. Pimm. 1976. Competition and niche shifts of mice introduced onto small islands. Oikos 27:251–258.
Forsberg, C. 1965. Environmental conditions for Swedish charophytes. Symbolae Botanicae Upsaliens 18:5–62.
Hicks, L.E. 1932. Ranges of pH-tolerance of the Lemnaceae. Ohio J. Sci. 32:115–131.
Kramer, P.J. 1969. Plant and soil water relationships. 482 p. McGraw-Hill, New York, N.Y.
Krebs, C.J. 1972. Ecology: the experimental analysis of distribution and abundance. 694 p. Harper and Row, New York, N.Y.
Lee, C. H. 1912. An intensive study of the water resources of a part of Owens Valley. USDI Geological Survey Water Supply Paper 294.
Littlefield, L., and C. Forsberg. 1965. Absorption and translocation of phosphorus-32 by Chara globularis Thuill. Physiol. Plantarum 18:291–296.
Lochard, R.B., and D.H. Owings. 1974. Moonrelated activities of bannertail (Dipodomys spectabilis ) and fresho (D . nitratoides ) kangaroo rats. Anim. Behav. 22:262–273.
Mason, H.L. 1957. A flora of the marshes of California. 878 p. University of California Press, Berkeley.
McLay, C.L. 1974. The distribution of duckweed (Lemnaperpusilla ) in a small southern California lake: an experimental approach. Ecology 55:262–276.
McLay, C.L. 1976. The effect of pH on the population growth of three species of duckweed: Spirodelaoligorrhiza , Lemnaminor , and Wolffiaarrhiza . Freshwater Biol. 6:125–136.
M'Closkey, R.T. 1980. Spatial patterns in sizes of seeds collected by four species of heteromyid rodents. Ecology 61:486–489.
M'Closkey, R.T., and B. Fieldwick. 1975. Ecological separation of sympatric rodents (Peromyscus and Microtus ). J. Mamm. 56: 119–129.
Merino, J., F. Garcia Nova, and M. Sanchez Diaz. 1976. Annual fluctuation of water potential in the xerophytic shrubs of the Donana Biological Reserve (Spain). Oecol. Plant. 11:1–11.
Meserve, P.L. 1974. Ecological relationships of two sympatric woodrats in a California coastal sage community. J. Mamm. 55:442–447.
Mueller-Dombois, D., and H. Ellenberg. 1974. Aims and methods of vegetation ecology. 547 p. John Wiley and Sons, New York, N.Y.
Mulligan, H.F., and A. Baranowski. 1969. Growth of phytoplankton and vascular aquatic plants at different nutrient levels. Vehr. Intern. Verein. Limn. 17:802–810.
Mulligan, H.F., A. Baranowski, and R. Johnson. 1976. Nitrogen and phosphorus fertilization of aquatic vascular plants and algae in replicated ponds. I. Initial response to fertilization. Hydrobiol. 48:109–116.
Pearcy, R.W. 1974. Comparative photosynthetic and respiratory gas characteristics of Atriplexlentiformis (Torr.) Wats. in coastal and desert habitats. Ecology 55:1104–1111.
Price, M.V. 1978. The role of microhabitat in structuring desert rodent communities. Ecology 59:910–921.
Rosenzweig, M.L. 1973. Habitat selection experiments with a pair of coexisting heteromyid rodent species. Ecology 54:111–117.
Rosenzweig, M.L., and J. Winakur. 1969. Population biology of a desert rodent community: habitat and environmental complexity. Ecology 50:558–572.
Schroder, G.E., and M.L. Rosenzweig. 1975. Perturbation analysis of competition and overlap in habitat utilizations between D . ordii and D . merriami . Oecol. 19:9–28.
Seddon, B. 1972. Aquatic macrophytes as limnological indicators. Freshwater Biol. 2:107–130.
Slatyer, R.O. 1967. Plant-water relationships. 366 p. Academic Press Inc., New York, N.Y.
Smith, R.L. 1966. Ecology and field biology. 686 p. Harper and Row, New York, N.Y.
Smith, R.L. 1974. Ecology and field biology. 850 p. Harper and Row, New York, N.Y.
Spence, D.H.N. 1967. Factors controlling the distribution of freshwater macrophytes with particular reference to the lochs of Scotland. J. Ecology 55:147–170.
Stickel, L.F. 1954. A comparison of certain methods of measuring ranges of small mammals. J. Mamm. 35:1–15.
Stickel, L.J. 1960. Peromyscus ranges at high and low population densities. J. Mamm. 41: 433–441.
Strickland, J.D., and T.R. Parsons. 1972. A manual of seawater analysis (second edition). 310 p. Bull. Fish. Res. Bd. Canada No. 167.
Whitford, W.G. 1976. Temporal fluctuations in density and diversity of desert rodent populations. J. Mamm. 57:351–369.
Williams, P.B. 1978. Changes in the Owens Valley shallow groundwater levels from 1970 to 1978. Inyo County Board of Supervisors.
Wondolleck, J.T. 1978. Forage-area separation and overlap in Heteromyid rodents. J. Mamm. 59:510–518.
Wood, R.D. 1950. Stability and zonation of Characeae. Ecology 31:642–647.
Zar, J.H. 1974. Biostatistical analysis. 620 p. Prentice-Hall, Inc., Englewood Cliffs, N.J.